Phage Therapy Unit of the Medical Centre of the Institute of Immunology and Experimental Therapy PAS
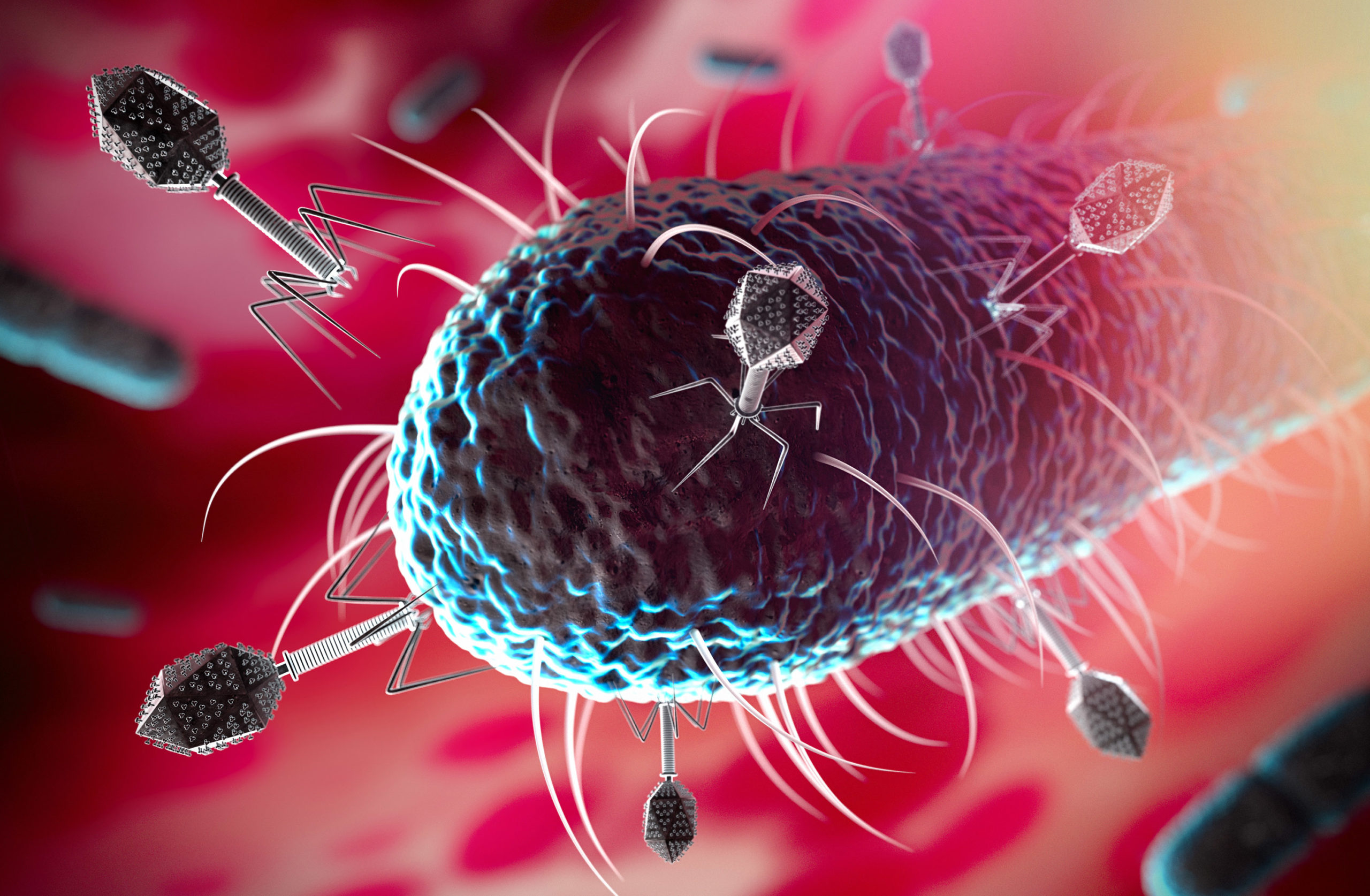
 Phage therapy is a method of treating bacterial infections which uses unique features of bacteriophages – bacterial viruses which attack only bacterial cells. Bacteriophages (or simply “phages”) are able to destroy different bacteria including those which are resistant to antibiotics and which cause life-threatening infections. It is this exceptional feature which determines the potential of phages in treating bacterial infections. This method had been known since the beginning of the 20th century.
Phage therapy is a method of treating bacterial infections which uses unique features of bacteriophages – bacterial viruses which attack only bacterial cells. Bacteriophages (or simply “phages”) are able to destroy different bacteria including those which are resistant to antibiotics and which cause life-threatening infections. It is this exceptional feature which determines the potential of phages in treating bacterial infections. This method had been known since the beginning of the 20th century.
The Institute of Immunology and Experimental Therapy PAS in Wroclaw (IIET PAS) has been conducting research on the biological properties and the application of bacteriophages for several decades. It has at its disposal methods for isolating bacteriophages and for preparing phage formulations which have been provided for different hospitals in Poland for phage therapy coordinated by the institute since the 1970s. To enable the application of phage therapy after Poland joined the European Union according to current regulations the institute opened its own Phage Therapy Unit (PTU) in 2005. Its aim is the admission of patients for treatment and its provision according to the protocol approved by an independent bioethics committee.
The Phage Therapy Unit is supported by the Bacteriophage Laboratory of the IIET PAS, which carries out phage typing procedures, prepares the phage formulations for patients and performs some other tests within experimental phage therapy. Recently two branches of the Phage Therapy Unit were opened in Cracow and Czestochowa. Since the results of standard clinical trials of phage preparations enabling their registration are not available yet, this form of treatment is currently possible only under the rules of a therapeutic experiment (on the basis of the respective Polish regulations) and in accord with the Declaration of Helsinki, also regulated in Poland by the Medical Profession Act of 5th December 1996. World Medical Association Declaration of Helsinki – Ethical Principles for Medical Research Involving Human Subjects: “ 37. In the treatment of an individual patient, where proven interventions do not exist or other known interventions have been ineffective, the physician, after seeking expert advice, with informed consent from the patient or a legally authorised representative, may use an unproven intervention if in the physician’s judgement it offers hope of saving life, re-establishing health or alleviating suffering. This intervention should subsequently be made the object of research, designed to evaluate its safety and efficacy. In all cases, new information must be recorded and, where appropriate, made publicly available.” The phage therapy is conducted on an outpatient basis under the protocol of an experimental programme: “Experimental phage therapy of drug-resistant bacterial infections, including MRSA infections” approved by an Independent Bioethics Committee (opinion No. KB-349/2005 – for English translation please click here). The programme is supervised by Professor Andrzej Górski, MD, PhD – the head of the Phage Therapy Unit and the Bacteriophage Laboratory. More information about our programme of experimental phage therapy is presented in Phage therapy basis and FAQs.
Contact to the PTU
Information:
Available in Polish or English on working days during 8:15 am – 3:45 pm.
Tel.: +48 71 370 99 01
E-mail: otf@hirszfeld.pl
Patient consultations and registration:
Phone registration is available on working days during 8:15 am – 3:45 pm.
Tel.: +48 71 370 99 01
Patient consultations in Polish or English by physicians are provided on working days during 8:00 am – 4:00 pm (Wroclaw, Czestochowa) and 3:00 pm – 7:00 pm (Cracow) only after previous registration.
Head of the Phage Therapy Unit:
Prof. Andrzej Górski MD, PhD
Tel.: +48 71 337 11 72
E-mail: agorski@ikp.pl
Phage Therapy Unit (main office in Wroclaw)
Medical Centre of the Institute of Immunology and Experimental Therapy PAS
Weigla 12
53-114 Wrocław, Poland
Tel.: +48 71 370 99 01 (direct) or +48 71 337 11 72 (institute’s reception)
Fax: +48 71 337 13 82
E-mail: otf@hirszfeld.pl
Location on a map
Phage Therapy Unit (branch in Cracow)
Medical Centre of the Institute of Immunology and Experimental Therapy PAS
Konecznego 2/4U
31-216 Kraków, Poland
Tel.: +48 12 420 60 70 (the number may be temporarily operated by an automatic answering machine)
Fax: +48 12 420 60 71
E-mail: otf.krakow@iitd.pan.wroc.pl
The rooms are located in the CAMI Clinic of Plastic Surgery and Laser Therapy (in Polish: Klinika Chirurgii Plastycznej i Laseroterapii „CAMI”) in Cracow
Location on a map
Phage Therapy Unit (branch in Czestochowa)
Medical Centre of the Institute of Immunology and Experimental Therapy PAS
Wały Dwernickiego 43/45
42-202 Częstochowa, Poland
Tel./fax: +48 34 534 50 00
E-mail: otf.klara@iitd.pan.wroc.pl
The rooms are located in the Klara Medical Center (in Polish: Centrum Medyczne Klara) in Czestochowa
Location on a map
Bank account for international money transfers:
Recipient:
Institute of Immunology and Experimental Therapy PAS
Weigla 12
53-114 Wroclaw, Poland
Recipient’s account data:
Bank Gospodarstwa Krajowego, Oddział we Wrocławiu
Malarska 26
50-111 Wrocław, Poland
IBAN number for transfers in Polish zloty: PL 84 1130 1033 0018 8181 8920 0001
IBAN number for transfers in euro: PL 92 1130 1033 0018 8181 8920 0051
SWIFT: GOSKPLPW
Head
Head: Prof. dr hab. n. med. Andrzej Górski, internal medicine specialist – clinical immunology. ORCID: https://orcid.org/0000-0002-0863-6041
Prof. Andrzej Górski had been working at the Institute since 1999. After his first term as rector of the Medical Academy in Warsaw he decided to apply for a post of a director of the Institute. As a head of Bacteriophage Laboratory he leads the team engaged in unique research on bacteriophages. The results of those studies have been published in reputed journals opening new perspectives for the therapeutic applications of phages. His initiative led to the establishment in 2005 of unique Phage Therapy Center treating patients with antibiotics-resistant infections with experimental phage therapy. Prof. A. Górski is also editor in chief of AITE, an Institute’s journal that became the leading scientific journal in Poland with IF of 3,200 (2019). Prof. A. Górski has also been active in the field of ethics in science that led to his election as chair, Commission for Ethics in Science (2019).
Current research
- Phage procurement and characterization for phage bank.
- Studies on the significance of anti-phage antibodies in phage therapy.
- Work on outlines of clinical trials on phage therapy.
Most important scientific achievements
- Editing of two multiple autor monographs on phage therapy that received high evaluations in The Lancet Infectious Diseases i Clinical Infectious Diseases:
- Borysowski J, Międzybrodzki R, Górski A. (2014). Phage Therapy: Current Research and Applications. Caister Academic Press (ISBN: 978-1-908230-40-9). Book review available at:
– https://academic.oup.com/cid/article/61/1/141/340829?searchresult=1
– https://www.thelancet.com/journals/laninf/article/PIIS1473-3099(14)70867-9/fulltext
- Górski A, Międzybrodzki R, Borysowski J. (2019). Phage Therapy: A Practical Approach. Springer International Publishing, (ISBN: 978-3-030-26736-0). Book review available at: – https://www.thelancet.com/journals/laninf/article/PIIS1473-3099(20)30307-8/fulltext
- Demonstration that production of anti-phage antibodies in patients on phage therapy depends on phage type, route and time of administration as well as patient’s conditions and is not correlated with the outcome of therapy.
- Demonstration that phages have immunomodulatory action which broadens their potential application in medicine. Hypothesis on the potential of phage therapy in COVID-19.(Górski A. et al. Phages in the fight against COVID-19? Future Microbiology 2020 vol. 15: 1095-1100. doi:10.2217/fmb-2020-0082; Górski A. et al. Sepsis, phages and COVID-19, Pathogens 2020, 9(10), 844; https://doi.org/10.3390/pathogens9100844).
Research methods
- Adam’s method to determine phage lytic activity.
- Gratia method to study RTD (routine test dilution).
- Colorimetric method to detect phage presence in biological and environemnal samples.
- ELISA mehod to determine anti-phage antibodies in patients treated with phage therapy.
- Bacteriophage purification using chromatography methods.
- In vitro tests on cell cultures.
- Statistical analysis of clinical data of patients treated with phages.
Key equipment
- Microbiological analyser VITEK 2 – compact BioMerieux
- Co2 incubator, Galaxy 48 New Brunswick
- Apparatus for anaerobic bacteria culturing Whitley H 35 HEPA
- Cell and phage counter ProtoCOL SYNBIOSIS
- Spectrophotometer Eppendorf
- Laminar flow unit S@peflow 1.2. BIOAIR
- Laminar flow unit CLAS II NUAIRE
- ELISA counter
- Light microscope with digital camera Leica
- High speed centrifuge Sigma 3- 30 KS
Main research projects
- „Studies on the immunomodulatory effects of bacteriophages on functions of immune cells, National Science Centre, 2018/31/B/NZ6/03999 OPUS 16, date of implementation:09.2019 – 11.09.2022, Head: Dr hab. Ryszard Międzybrodzki. The project budget: 1 499 000 zł.
- Database of Scientific Information Supporting Innovative Therapies (BINWIT), European Funds Digital Poland, 02.03.01-IP.01-00-006/17, date of implementation: 01.08.2018 – 31.07.2021, Head: dr Krzysztof Jakub Pawlik, Head of the Bacteriophages Team – Prof. Andrzej Górski. The project budget: 18 867 895 zł.
- „Antiviral effects of phages”, National Science Centre, 2013/11/B/NZ1/02107, OPUS 6, date of implementation :20.08.2014 – 19.08.2018, Head: Prof. Andrzej Górski. The project budget: 929 600 zł.
- „Optimization of the characteristics and preparation of phage preparations for therapeutic purposes”, The National Centre for Research and Development, Operational Programme Innovative Economy, POIG.01.03.01-02-003/08, date of implementation: 01.09.2007 – 31.08.2014, Head: Andrzej Górski. The project budget: 13 660 000 zł.
- „Innovative bacteriophage protective preparation for use in the diabetic foot syndrome”, The National Centre for Research and Development, Operational Programme Innovative Economy, POIG.01.03.01-02-048/12, date of implementation:04.2013 – 31.12.2015, Head: Dr Beata Weber-Dąbrowska. The project budget: 9 137 820 zł
Selected publications
- Górski A, Międzybrodzki R, Żaczek M, Borysowski J. Phages in the fight against COVID-19? Future Microbiol. 2020 Aug 26:10.2217/fmb-2020-0082. doi: 10.2217/fmb-2020-0082. Epub ahead of print. PMID: 32845164; PMCID: PMC7451411.
- Górski A, Międzybrodzki R, Węgrzyn G, Jończyk-Matysiak E, Borysowski J, Weber-Dąbrowska B. Phage therapy: Current status and perspectives. Med Res Rev. 2020 Jan;40(1):459-463. doi: 10.1002/med.21593. Epub 2019 May 7. PMID: 31062882.
- Górski A, Międzybrodzki R, Jończyk-Matysiak E, Żaczek M, Borysowski J. Phage-specific diverse effects of bacterial viruses on the immune system. Future Microbiol. 2019 Sep;14(14):1171-1174. doi: 10.2217/fmb-2019-0222. Epub 2019 Sep 19. PMID: 31535921; PMCID: PMC6802706.
- Górski A, Bollyky PL, Przybylski M, Borysowski J, Międzybrodzki R, Jończyk-Matysiak E, Weber-Dąbrowska B. Perspectives of Phage Therapy in Non-bacterial Infections. Front Microbiol. 2019 Jan 9;9:3306. doi: 10.3389/fmicb.2018.03306. PMID: 30687285; PMCID: PMC6333649.
- Górski A, Jończyk-Matysiak E, Łusiak-Szelachowska M, Międzybrodzki R, Weber-Dąbrowska B, Borysowski J. Bacteriophages targeting intestinal epithelial cells: a potential novel form of immunotherapy. Cell Mol Life Sci. 2018 Feb;75(4):589-595. doi: 10.1007/s00018-017-2715-6. Epub 2017 Nov 21. PMID: 29164271; PMCID: PMC5769817.
- Borysowski J, Ehni HJ, Górski A. Ethics review in compassionate use. BMC Med. 2017 Jul 24;15(1):136. doi: 10.1186/s12916-017-0910-9. PMID: 28735571; PMCID: PMC5523146.
- Majewska J, Kaźmierczak Z, Lahutta K, Lecion D, Szymczak A, Miernikiewicz P, Drapała J, Harhala M, Marek-Bukowiec K, Jędruchniewicz N, Owczarek B, Górski A, Dąbrowska K. Induction of Phage-Specific Antibodies by Two Therapeutic Staphylococcal Bacteriophages Administered per os. Front Immunol. 2019 Nov 14;10:2607. doi: 10.3389/fimmu.2019.02607. PMID: 31803179; PMCID: PMC6871536.
- Górski A, Międzybrodzki R, Borysowski J (Eds.), Phage Therapy: A Practical Approach, Springer International Publishing (2019). ISBN: 978-3-030-26736-0, doi: 10.1007/978-3-030-26736-0.
- Borysowski J, Międzybrodzki R, Górski A. (Eds.). Phage Therapy: Current Research and Applications. Caister Academic Press (2014). ISBN: 978-1-908230-40-9.
 Phage therapy is a method of treating bacterial infections which uses unique features of bacteriophages – bacterial viruses which attack only bacterial cells. Bacteriophages (or simply “phages”) are able to destroy different bacteria including those which are resistant to antibiotics and which cause life-threatening infections. It is this exceptional feature which determines the potential of phages in treating bacterial infections. This method had been known since the beginning of the 20th century.
Phage therapy is a method of treating bacterial infections which uses unique features of bacteriophages – bacterial viruses which attack only bacterial cells. Bacteriophages (or simply “phages”) are able to destroy different bacteria including those which are resistant to antibiotics and which cause life-threatening infections. It is this exceptional feature which determines the potential of phages in treating bacterial infections. This method had been known since the beginning of the 20th century.